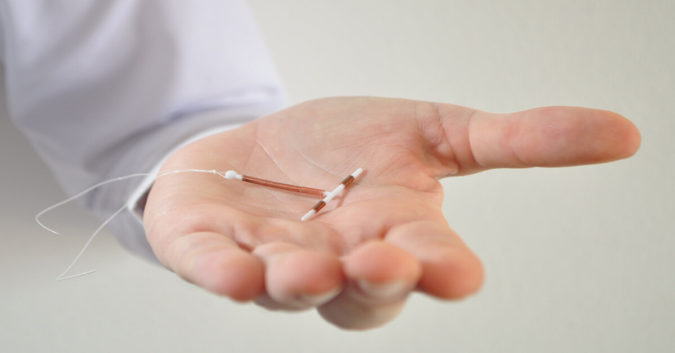
Paragard® is a copper intrauterine device (IUD) used by thousands of American women to provide long-term birth control. It is one of five available IUDs on the United States’ birth control market and the only one that is completely hormone-free. Given the lack of hormones, the device is quite popular.
Despite its popularity, a number of recent lawsuits allege a concerning manufacturing flaw in this small piece of plastic and copper. Dozens of women are claiming that their Paragard IUD broke before or during its removal, leaving behind chunks of copper or plastic lodged in the uterus or another organ.
To state the obvious, this is a problem. Leftover materials, whether plastic or copper, can lead to long-term health complications, such as pain and infertility. In some cases, women have required surgery to remove the broken pieces of the Paragard IUD.
Prior to 2019, the Makers of Paragard Offered No Warning
In legal filings, plaintiffs say they were not adequately warned of these risks and are seeking compensation to cover the damages.
These allegations follow a 2019 ruling by the U.S. Food and Drug Administration (FDA), which ordered Teva Pharmaceuticals, the makers of Paragard, to update the device’s label to include a warning that the device can get lodged inside of the body and break.
While Paragard now includes instructions for healthcare providers on how to remove the IUD, and mentions “device breakage” as a possible adverse event, it does not state how often such breakages may occur.
Specifically, Teva warns that the IUD’s copper-lined plastic threads may “retract into the uterus or break,” or it can “break, perforate the uterus, or be expelled.”
While vague, the update may prove helpful to women who are currently considering the IUD as a birth control option. The problem is that, for many women who have been using Paragard for years, the information comes much too late.
The device has been in use since the 1980s and is designed to remain inserted for up to 12 years.
How Dangerous Is Paragard?
Much of Paragard’s popularity is owed to its lack of hormonal side-effects, especially when compared to common estrogen- and progestin-based birth control alternatives.
According to adverse reports and Paragard lawsuits, the most dangerous side-effect has to do with the way the device is built, and the elevated risk of its removal. What’s tragic is that this risk was hidden from patients for years, barring them from making truly informed decisions with an accurate understanding of the device’s risks.
That Paragard may break before or during its removal is a significant enough risk to have deterred some women from ever using it. But those women were denied critical information and, through no fault of their own, may suffer injury as a result. They should not have to pay for any such injuries.
Worse yet, if a doctor is not able to retrieve the remains of a broken IUD, the patient may endure long-term pain and suffering, infertility, or organ problems, requiring surgery that may or may not be covered by their health insurance.
Such circumstances could have and should have been avoided. It should come as no surprise, then, that more than 80 Paragard lawsuits are currently pending, and lawyers expect many more to be filed in the coming months.
If a Paragard IUD device broke inside your body before or during removal in the last 3 years, contact Sokolove Law for a free consultation. Our law firm has won over $1.7 Billion for victims of defective medical devices, and we may be able to help you take legal action.
Our Paragard IUD lawsuit attorneys will review your case for free.
All brands are trademarks of their respective companies.